Prototype of running clinical trials in an untrustworthy environment using blockchain | Nature Communications

Development of a Multi-Centre Clinical Trial Data Archiving and Analysis Platform for Functional Imaging | Semantic Scholar

Sharing and reuse of individual participant data from clinical trials: principles and recommendations | BMJ Open
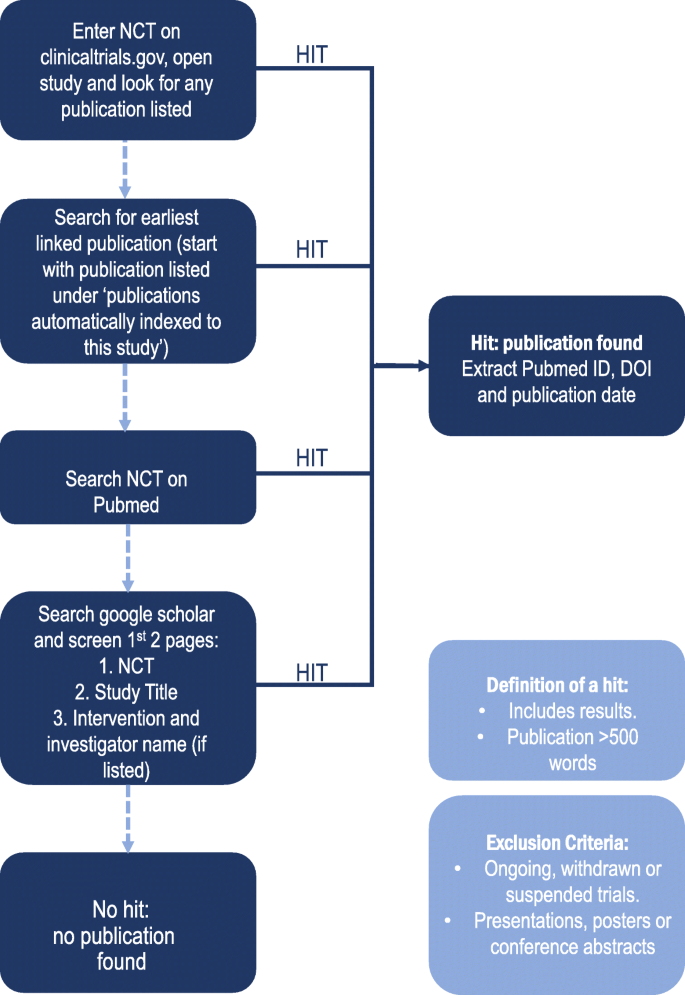
Missing clinical trial data: the evidence gap in primary data for potential COVID-19 drugs | Trials | Full Text

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet